
Fossil fuel combustion creates numerous harmful products. When these products are released to the environment, they endanger both humans and the environment. One class of environmentally hazardous products created during fossil fuel combustion is oxides of nitrogen. These products, which are collectively known as “NOx”, contribute to the environmental problems of acid rain and the formation of atmospheric ozone and smog.
There are five chemical compounds which are defined as NOx. These are:
Nitric oxide (NO) and nitrogen dioxide (NO2) are the most common forms of NOx created in the fossil fuel combustion process. NO is the primary chemical formed in the combustion process. The NO generally oxidizes to form NO2.
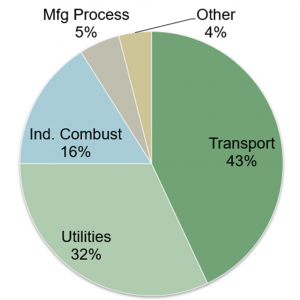
The global emissions of NOx into the atmosphere have been increasing steadily since the middle of the last century. A significant amount of increased emissions is attributed to human activities, in particular to increased combustion of biomass, such as wood, trash and other organic materials, and fossil fuels.
NOx is characterized by the source of nitrogen in the combustion process which contributes to the formation of NOx. When the source of nitrogen is the fuel, it is referred to as “fuel NOx.” When the source of nitrogen is air, it is referred to as “thermal NOx.”
There are numerous industrial sources of NOx. They include the following industries:
NOx has the following environmental and health impacts:
Nitrogen Dioxide (NO2):
Atmospheric Ozone & Smog:
Acid Rain:
NOx is created in virtually all fossil fuel combustion processes. Knowledgeable operation of your power plant can lead to increased efficiency and a decrease in environmental pollutants. FCS offers Boiler Operation for Environmental Compliance and Efficiency, which fulfills the State of Maryland’s requirements for Operator Training for NOx Compliance.