
Carbon and our future.
Carbon (specifically carbon dioxide) has been a hot topic for the past few decades. In recent years, carbon has become the misunderstood villain of the climate change story. How can carbon, which exists in every living thing, possibly be bad? In this blog, we’re going to “crack” open carbon and see if all the negative publicity is valid.
Carbon (chemical symbol C) is the fourth most abundant element in the universe by mass, and the 15th most abundant element in the Earth’s crust[1]. Due to its abundance and ability to form many different bonds, carbon is found in all forms of life on Earth, and makes up about 50% of the dry biomass (material produced by living things)[2].
If carbon is already all around us, why has it been targeted recently? One specific carbon bond is to blame: carbon dioxide (CO2). Carbon dioxide is a naturally occurring gas (1 carbon atom and 2 oxygen atoms), famously given off when we breathe. Carbon dioxide is a “greenhouse gas,” a gas that absorbs and radiates heat. This reflection of heat back onto the Earth’s surface is the major contributor to “global warming.”
Carbon Dioxide in the atmosphere is necessary. Oxygen and nitrogen (which make up most of the atmosphere), don’t reflect ANY considerable heat back onto the Earth. Without CO2, Earth’s average temperature would be below freezing[3].
Researchers have been able to determine the average amount of Carbon Dioxide in the atmosphere over the last 800,000 years (Figure 1). During the last Ice Age (about 20,000 years ago) CO2 levels were around 180 parts per million (ppm) and the average global temperature was around 46 degrees (Fahrenheit). Today, levels are around 420 ppm and the average temperature is 59 degrees.
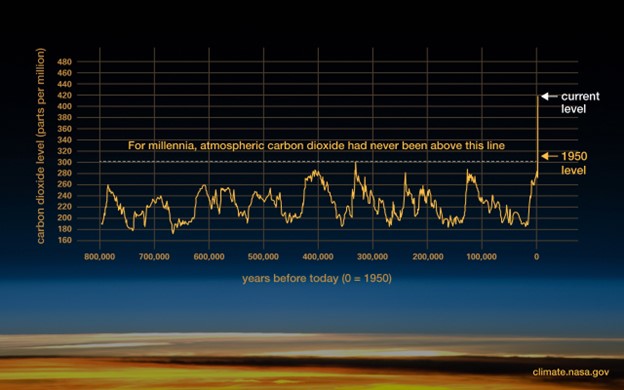
Figure 1 – Carbon Dioxide in the Atmosphere for the Previous 800,000 Years. (courtesy NASA)
Some people argue these changes in temperature are natural. The research in Figure 1 does show a relatively predictable rise and fall in CO2 1950, when CO2 levels began to rise rapidly. Looking at almost a million year’s worth of data, wouldn’t it be safe to assume that this trend will also turn and lead us back down into another Ice Age?
You probably remember from science class, or Bill Nye, that matter can’t be created, or destroyed. We have just as many atoms of carbon now as we did 800,000 years ago, so why the sudden spike?
The answer is the Carbon Cycle (Figure 2). This process moves carbon around our world. As the Department of Energy explains, Earth does not lose carbon, carbon just moves from place to place. In the “Natural Carbon Cycle,” CO2 in the atmosphere is absorbed by ocean and plants, where natural processes utilize it to create something new.
Living creatures return carbon to the air when they breathe and return some to the soil when they die and decompose. Throughout history, this cycle has maintained a relative balance in atmospheric carbon. So, what changed? Human activity has negatively impacted the balance of the carbon cycle in two major ways: Land usage and fossil fuels.
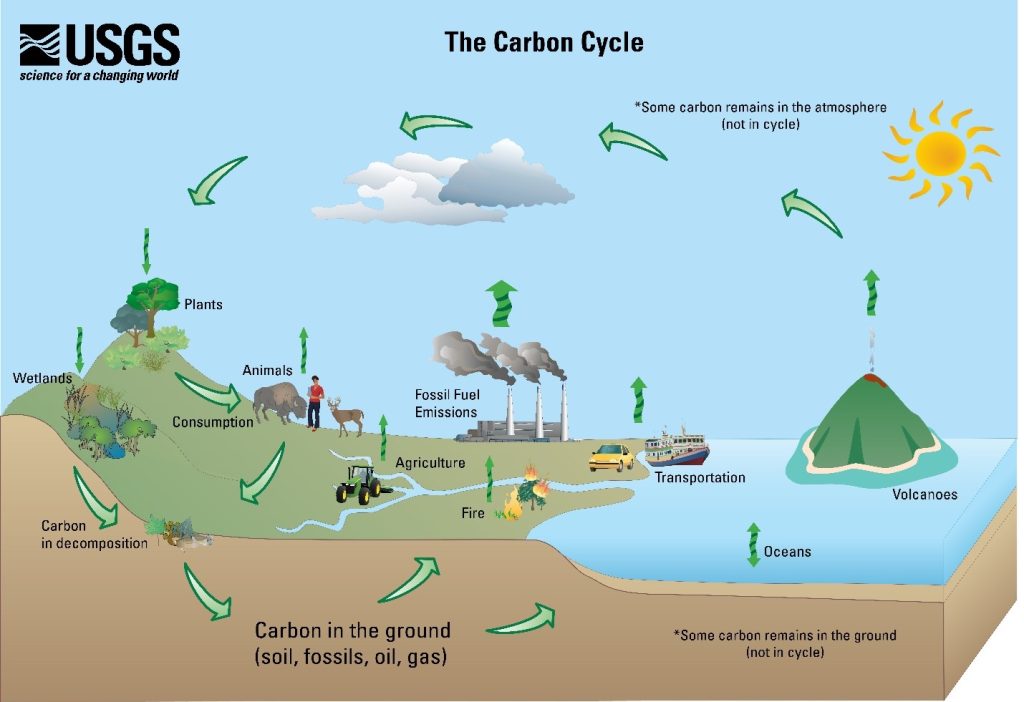
Figure 2 – The Carbon Cycle (Courtesy USGS)
During the Industrial Revolution, humans became reliant on the burning of fossil fuels to create a new way of life. Electricity, cars, manufacturing, all heavily reliant on high carbon content fuels. We have been releasing into the atmosphere carbon that took nature MILLIONS of years to absorb.
The increased quality of life also resulted in a population boom. According to the World Economic Forum Since 1800, the world’s population has increased 800%. That population increase came with mass deforestation and led to industrial level farming.
Mass deforestation removes the number one natural carbon storage unit: trees. For example, the General Sherman sequoia in California holds 2.2 million pounds of carbon!
An article from the National Institutes of Health (NIH) explains that carbon absorbed in the soil is released every time that soil is “opened” and explores ways in which we can change our methods of farming to take advantage of the carbon storing capabilities of soil. Microorganisms in the soil absorb carbon and use it to grow, which makes the soil healthy.
Carbon is also absorbed by the ocean. As the ocean absorbs more carbon in the world’s attempt to chemically balance itself, the pH of the ocean lowers, which makes it more acidic. It also causes temperatures to change, and can actually affect the currents in the ocean, leading to extreme weather shifts.
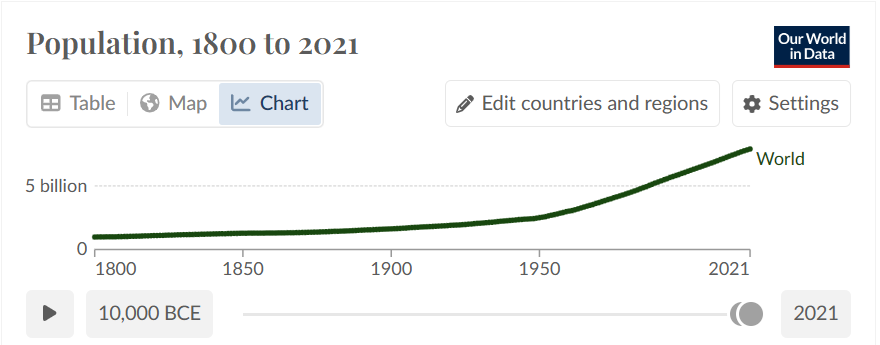
Figure 3 – World Population in the Past 221 years.
Excess CO2 is not all bad though right? According to NASA, the Earth is greener than ever over the last 20 years. While some argue that is proof there is no problem, scientists equate the increase in foliage mainly to better land use practices and reforestation efforts, not an increase in atmospheric plant food and warmer temperatures.
Ok, let’s recap what we know so far:
So, what can we do differently? Until recently, the only solution presented has been to reduce emissions. While we all agree that reducing emissions is a good idea, the methods and costs (both financial and human) have caused a lot of debate.
Now that we understand how the natural carbon cycle works, scientists are working on ways to boost that natural cycle. In a process called carbon sequestration carbon is absorbed either during the source process or from the atmosphere and “sequestered” or stored in another form.
So why are we not all in on these processes? Every process has pros and cons. It is important to understand these pros and cons, and to ensure we do not just create a new problem. Let’s take a look at some options.
Emissions reduction is the current top contender for climate change champion, and for good reason. We know that the fuels we burn for energy and transportation are releasing carbon that took millions of years to remove from the atmosphere. According to the Intergovernmental Panel on Climate Change (IPCC), we need to achieve “Net-Zero” carbon emissions by 2050 in order to “cap” global warming at 1.5 degrees (Celsius).
How can we go about achieving Net-Zero emissions? If you follow the news, you are well aware that there is a “war” on fossil-fueled power plants (coal particularly) and the Internal Combustion Engine. As shown in Figure 4, Transportation and Electrical Generation are over half of the contribution to CO2 emissions.
In both transportation and power generation, renewable sources have not yet reached a point where they can replace traditional sources with the same reliability. This is in part because we have become accustomed to a certain way of life. It is not uncommon for Americans to drive an hour or more to work each day. Our commerce relies heavily on long-haul trucking. Large rural expanses preclude the use of nationwide mass transit.
The race to decarbonize both sectors at once is leading to further issues, as discussed in Rick Cragg’s blog, simultaneously adding more electric cars to the road and reducing the number of large base load power plants is causing problems with grid stability. Famously, California had to request people refrain from charging their vehicles during a heat wave to prevent rolling blackouts. In the north, EV owners were struggling to charge their vehicles during the extreme cold.
Renewable energy projects are not cheap, and the upgrading of the infrastructure needed to wholesale replace base load plants will be an extensive project. A CO2 “free” solution to replacing large fossil fuel plants is to further the use of Nuclear energy. Nuclear energy has a bad reputation because of incidents like Chernobyl and Fukushima. However, construction of new nuclear power plants is also a costly venture.
Yes of course! However, someone has to pay for it. The challenge for those trying to reduce emissions is to find the right balance between economics and environmental outcomes. For an easy example, an F-150 Lightning starts at $49,995. Without any upgrades, and without the installation of the supercharger in your home. The comparable conventional F-150 starts at $40,780.
Scale this to the size of a commercial fleet of electric vehicles, or the cost comparison between building a gas boiler power plant or an equivalent sized windfarm, and you can understand the struggle to find financial backers. We compared the production and the human costs of renewable energy sources in a recent blog.
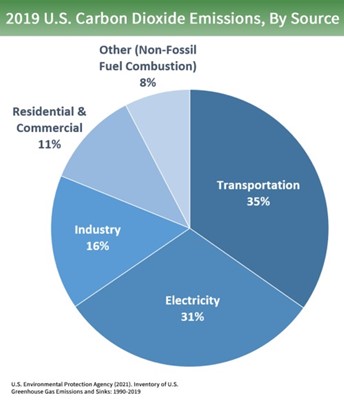
Figure 4 – Carbon Dioxide Emissions by Source (courtesy US EPA)
The largest pro is that technology is improving. The National Renewable Energy Laboratory (NREL) released a report discussing how technology can be adjusted to both reduce the need for traditional inertial energy for grid stability, and to provide an alternative source of stability using renewables.
Even the technology used in renewable energy equipment is improving, solar panels and wind turbines are becoming more efficient, and we’re getting smarter about where to use them. Projects like the Agrivoltaics research being done at the University of Illinois are finding ways to utilize the land around solar panels, so that the land is not “wasted” as many people now consider it to be.
Technology is also becoming more affordable. As processes improve and the market scales up, the process is becoming more efficient.
There are many known alternatives to fossil energy, or to high emissions producing fossil fuel use. Natural gas emits nearly half the CO2 of coal, and something as simple as building hybrid vehicles drastically reduces emissions from transportation.
Finally, emissions reduction is the part of the puzzle everyone can do something about, and many of the steps we can individually take to reduce the emissions we cause also save us money. Reducing unnecessary driving, carpooling, saving electricity, are all small steps we can take to reduce the rate at which we are bypassing the carbon cycle.
Carbon capture is a technique used to take the carbon released from a process and return it to another part of the cycle instead of going to the atmosphere. There are two categories of carbon capture being studied: Natural and Technological.
Natural carbon capture looks to the way nature already captures carbon, either geologically or biologically. The biggest Pro? Many of the natural processes are simple and provide benefits to the environment beyond capturing CO2. Restoring and protecting wetlands, re-forestation, and improving technologies to produce reliable, high yield crops using more natural processes (No-Till Farming) are all ways of naturally capturing carbon. Basically, this method just expands the use of what nature already does.
Other methods of “Natural” carbon capture involve synthetically boosting the natural processes. These processes include Enhanced Weathering, or grinding existing rocks up, so that the natural process of minerals mixing with CO2 to form new rocks is also sped up. Other forms include Direct Ocean Carbon Capture, where the CO2 is pulled from the ocean water and converted into a liquid, which causes the ocean to absorb more CO2 from the atmosphere.
First, like renewables, the technology is currently not scalable to the rate at which we need to remove CO2. The infrastructure and technology development will take time, and money before it can be constructed at scale. Energy will be required to power these processes, which may offset the benefit.
Finally, with any natural process, we have to make sure that correcting one problem does not create a new problem. One major concern with the new technology is the feasibility of long-term storage and the effect of failure. Current research shows promise in returning the captured carbon to the ground. This can happen using the same wells and mines we pulled the fossil fuels and coal from in the first place.
Technological carbon capture utilizes industrial processes to extract carbon dioxide from a point source. For example the exhaust stack of a coal boiler, or from a refinery.
This process, often referred to as Carbon Capture and Storage, or CCS, uses chemicals and equipment to capture, separate, compress, and transport the CO2. Carbon Dioxide removal has been used in both the natural gas industry and in space and underwater exploration for years, so one Pro is that technology already exists, and can be scaled.
In some processes, like Direct Air Capture (DAC), air is drawn through the system and the CO2 is extracted directly from the air. It is then converted utilized in another process or converted into a liquid or solid for storage. Current uses for CCS include:
The biggest Cons with technological carbon capture are similar to the cons associated with the point sources they are designed for. These installations require extensive infrastructure (land and electrical power) and often require large amounts of resources (chemicals and/or water) to perform their function.
The World Resources Institute has great information on the scale needed to reverse current CO2 emissions and what that currently costs in resources. For example, current systems can use as much as 7 tonnes of water per tonne of CO2 removed. With water already considered scarce in many locations, this is a major hurdle.
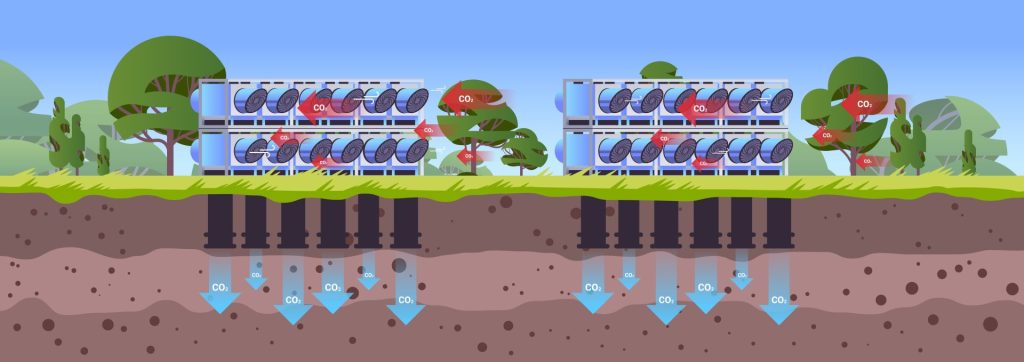
Figure 5 – Decarbonization carbon dioxide capture and storage
While the last 100 years may have seen humans cause a lot of damage to the environment, the last 20 – 30 years have seen huge improvements in the technologies and preservation of resources we had previously taken for granted. As we move forward, we have to be smart about the methods we use. By working WITH nature, setting realistic goals, and improving the way we manage resources, we can successfully balance quality of life and take care of
[1] Anderson, Don L.; “Chemical Composition of the Mantle”, Theory of the Earth, pp. 147–175 ISBN 0865421234
[2] 5.1 Carbon Content of Vegetation. 2015 Knowledge reference for national forest assessments, Food and Agriculture Organization of the United Nations, https://openknowledge.fao.org/server/api/core/bitstreams/dd5e7109-24ba-4494-8138-f2d00f810e7e/content